Why Peptide Purity and HPLC Testing Matter in Research
Introduction
In laboratory research, molecular precision is critical.
When working with peptides, even minor impurities can influence receptor binding, signalling behaviour, and experimental outcomes.
That is why purity verification and analytical testing are central to responsible peptide research.
All products supplied by CoreLab Supplies include a Certificate of Analysis (COA) specific to each batch. We also implement internal QA/QC procedures, including periodic independent third-party testing to verify quality standards
What Does Peptide Purity Mean?
Peptide purity refers to the percentage of the desired amino acid sequence present within a batch.
During synthesis, by-products can occur, including:
Truncated sequences Deletion variants Oxidised residues Side-reaction compounds
Higher purity reduces the likelihood of unintended molecular interaction.
Why Small Impurities Can Matter
In research models, small variations may affect:
Binding affinity Signal intensity Receptor selectivity Assay reproducibility
While impurities may exist in trace amounts, they can still influence biological systems depending on experimental design.
For this reason, analytical verification is essential.
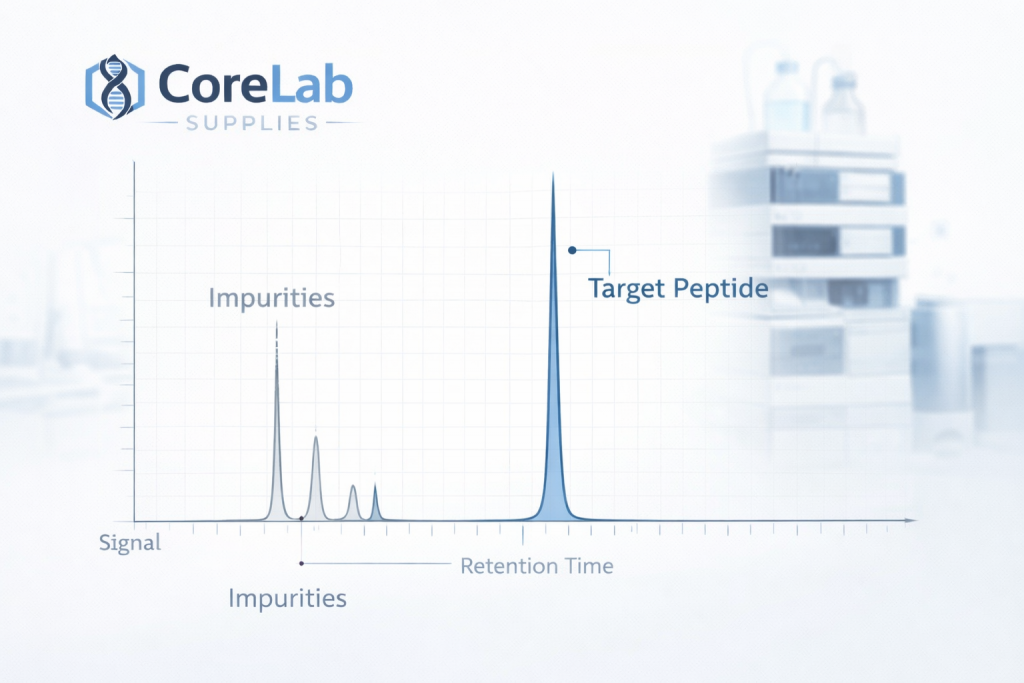
High Performance Liquid Chromatography (HPLC)
HPLC is one of the primary analytical methods used to determine peptide purity.
The process involves:
1 – Separating molecular components within a sample
2 – Measuring retention times
3 – Calculating relative peak areas
A dominant single peak typically represents the intended peptide sequence.
Minor peaks may represent impurities or synthesis by-products.
Mass Spectrometry Confirmation
While HPLC measures purity, mass spectrometry confirms molecular identity.
It verifies:
Correct molecular weight Sequence consistency Absence of major truncations
Together, these methods provide analytical confidence.
Why Batch Verification Supports Reproducibility
In scientific research, reproducibility is fundamental.
Consistent material quality ensures:
Reliable experimental results Reduced variability Greater confidence in data interpretation
Analytical documentation allows researchers to confirm material specifications prior to use.
The Role of Documentation
A Certificate of Analysis (COA) typically includes:
Batch number Purity percentage Analytical method Chromatogram reference Date of testing.
For example our latest Batch of Retatrutide 20mg – Purity 99.83% – Confirmed by third party testing
Documentation strengthens transparency within research supply chains.
Conclusion
Peptide research depends on precision.
Purity testing and analytical verification are not marketing extras — they are foundational to experimental integrity.
Understanding how peptides are analysed allows researchers to make informed decisions and maintain scientific standards.
FAQ
What purity level is typical for research peptides?
High-quality research material is often ≥98% purity, depending on synthesis method and application.
Does 100% purity exist?
Absolute purity is extremely difficult to achieve due to the nature of chemical synthesis.